يوديد الألومنيوم
| |

| |
| الأسماء | |
|---|---|
| اسم أيوپاك المفضل
Aluminium iodine | |
| أسماء أخرى
Aluminium(III) iodide
Aluminum iodide | |
| Identifiers | |
| رقم CAS | |
3D model (JSmol)
|
|
| ChemSpider |
|
| ECHA InfoCard | 100.029.140 |
| رقم EC |
|
PubChem CID
|
|
| UNII |
|
| UN number | UN 3260 |
CompTox Dashboard (EPA)
|
|
| InChI | InChI={{{value}}} |
| SMILES | |
| الخصائص | |
| الصيغة الجزيئية | AlI3, AlI3·6H2O (hexahydrate) |
| كتلة مولية | 407.695 g/mol (anhydrous) 515.786 g/mol (hexahydrate)[1] |
| المظهر | white (anhydrous) or yellow powder (hexahydrate)[1] |
| الكثافة | 3.98 g/cm3 (anhydrous)[1] 2.63 g/cm3 (hexahydrate)[2] |
| نقطة الانصهار | |
| نقطة الغليان | |
| قابلية الذوبان في الماء | very soluble, partial hydrolysis |
| قابلية الذوبان في alcohol, ether | soluble (hexahydrate) |
| البنية | |
| البنية البلورية | Monoclinic, mP16 |
| الزمرة الفراغية | P21/c, No. 14 |
| ثابت العقد | a = 1.1958 nm, b = 0.6128 nm, c = 1.8307 nm |
| ثابت العقد | α = 90°, β = 90°, γ = 90° |
| الكيمياء الحرارية | |
| الإنتالپية المعيارية للتشكل ΔfH |
-302.9 kJ/mol |
| Standard molar entropy S |
195.9 J/(mol·K) |
| سعة الحرارة النوعية، C | 98.7 J/(mol·K) |
ما لم يُذكر غير ذلك، البيانات المعطاة للمواد في حالاتهم العيارية (عند 25 °س [77 °ف]، 100 kPa). | |
| مراجع الجدول | |
يوديد ألمنيوم Aluminium iodide مركب كيميائي له الصيغة AlI3 ، ويكون على شكل بلورات عديمة اللون. [3] or the action of HI on Al metal. The hexahydrate is obtained from a reaction between metallic aluminum or aluminum hydroxide with hydrogen iodide or hydroiodic acid. Like the related chloride and bromide, AlI 3 is a strong Lewis acid and will absorb water from the atmosphere. It is employed as a reagent for the scission of certain kinds of C-O and N-O bonds. It cleaves aryl ethers and deoxygenates epoxides.[4]
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
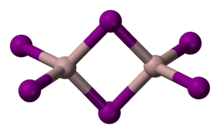
البنية
Solid AlI 3 is dimeric, consisting of Al 2I 6, similar to that of AlBr 3.[5] The structure of monomeric and dimeric forms have been characterized in the gas phase.[6] The monomer, AlI 3, is trigonal planar with a bond length of 2.448(6) Å, and the bridged dimer, Al 2I 6, at 430 K is a similar to Al 2Cl 6 and Al 2Br 6 with Al−I bond lengths of 2.456(6) Å (terminal) and 2.670(8) Å (bridging). The dimer is described as floppy with an equilibrium geometry of D2h.
يوديد الألومنيوم الأحادي
2(s) → 2AlI
3(s) is at the origin of the phenomenon observed.
The name "aluminium iodide" is widely assumed to describe the triiodide or its dimer. In fact, a monoiodide also enjoys a role in the Al–I system, although the compound AlI is unstable at room temperature relative to the triiodide:[7]
An illustrative derivative of aluminium monoiodide is the cyclic adduct formed with triethylamine, Al 4I 4(NEt 3) 4.
المصادر
- ^ أ ب ت Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, FL: CRC Press. p. 4.45. ISBN 1439855110.
- ^ Perry, Dale L. (19 April 2016). Handbook of Inorganic Compounds, Second Edition. CRC Press. p. 8. ISBN 978-1-4398-1462-8.
- ^ Watt, George W; Hall, James L; Taylor, William Lloyd; Kleinberg, Jacob (1953). "Aluminum Iodide". Inorganic Syntheses. Inorganic Syntheses. Vol. 4. pp. 117–119. doi:10.1002/9780470132357.ch39. ISBN 9780470132357.
- ^ Gugelchuk, M. (2004). "Aluminum Iodide". In L. Paquette (ed.). Encyclopedia of Reagents for Organic Synthesis. New York: J. Wiley & Sons. doi:10.1002/047084289X.ra083. ISBN 0471936235.
- ^ Troyanov, Sergey I.; Krahl, Thoralf; Kemnitz, Erhard (2004). "Crystal structures of GaX3(X= Cl, Br, I) and AlI3". Zeitschrift für Kristallographie. 219 (2–2004): 88–92. doi:10.1524/zkri.219.2.88.26320. S2CID 101603507.
- ^ Hargittai, Magdolna; Réffy, Balázs; Kolonits, Mária (2006). "An Intricate Molecule: Aluminum Triiodide. Molecular Structure of AlI3and Al2I6 from Electron Diffraction and Computation". The Journal of Physical Chemistry A. 110 (10): 3770–3777. doi:10.1021/jp056498e. PMID 16526661.
- ^ Dohmeier, C.; Loos, D.; Schnöckel, H. (1996). "Aluminum(I) and Gallium(I) Compounds: Syntheses, Structures, and Reactions". Angewandte Chemie International Edition. 35 (2): 129–149. doi:10.1002/anie.199601291.
وصلات خارجية
 Media related to يوديد الألومنيوم at Wikimedia Commons
Media related to يوديد الألومنيوم at Wikimedia Commons
| HI | He | ||||||||||||||||
| LiI | BeI2 | BI3 | CI4 | NI3 | I2O4, I2O5, I4O9 | IF, IF3, IF5, IF7 | Ne | ||||||||||
| NaI | MgI2 | AlI3 | SiI4 | PI3, P2I4 | S | ICl, ICl3 | Ar | ||||||||||
| KI | CaI2 | Sc | TiI4 | VI3 | Cr | MnI2 | Fe | CoI2 | NiI2 | CuI | ZnI2 | Ga2I6 | GeI2, GeI4 | AsI3 | Se | IBr | Kr |
| RbI | SrI2 | Y | ZrI4 | Nb | Mo | Tc | Ru | Rh | Pd | AgI | CdI2 | InI3 | SnI4, SnI2 | SbI3 | TeI4 | I | Xe |
| CsI | BaI2 | Hf | Ta | W | Re | Os | Ir | Pt | AuI | Hg2I2, HgI2 | TlI | PbI2 | Bi | Po | At | Rn | |
| Fr | Ra | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Uub | Uut | Uuq | Uup | Uuh | Uus | Uuo | |
| ↓ | |||||||||||||||||
| La | Ce | Pr | Nd | Pm | SmI2 | Eu | Gd | TbI3 | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac | ThI4 | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||
