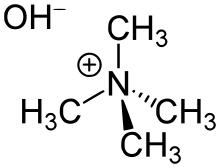
هيدروكسيد رباعي مثيل الأمونيوم
| |
| |
| الأسماء | |
|---|---|
| اسم أيوپاك
tetramethylazanium hydroxide
| |
| أسماء أخرى
tetramethylammonium hydroxide; N,N,N,-trimethylmethanaminium hydroxide
| |
| المُعرِّفات | |
| رقم CAS | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.000.803 |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| الخصائص | |
| الصيغة الجزيئية | C4H13NO |
| كتلة مولية | 91.15 g mol-1 |
| الكثافة | ~ 1.015 g/cm3 (20-25% aqueous solution) |
| نقطة الانصهار | |
| نقطة الغليان | |
| قابلية الذوبان في الماء | high |
| المخاطر | |
| صفحة بيانات السلامة | Sigma-Aldrich MSDS for TMAH·5H2O |
| ن.م.ع. مخطط تصويري |  
|
| ن.م.ع. كلمة الاشارة | Danger[1] |
| H300, H311, H314[1] | |
| P260, P264, P270, P280, P301+P310, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P322, P361, P363, P405, P501[1] | |
| NFPA 704 (معيـَّن النار) | |
| مركبات ذا علاقة | |
أنيونات أخرى
|
كلوريد رباعي مثيل الأمونيوم |
كاتيونات أخرى
|
tetraethylammonium hydroxide |
ما لم يُذكر غير ذلك، البيانات المعطاة للمواد في حالاتهم العيارية (عند 25 °س [77 °ف]، 100 kPa). | |
| مراجع الجدول | |
هيدروكسيد رباعي مثيل الأمونيوم (Tetramethylammonium hydroxide؛ TMAH أو TMAOH) هو quaternary ammonium salt with the molecular formula N(CH3)4+ OH−. It is commonly encountered as concentrated solutions in water or methanol. The solid and solutions are colorless, or yellowish if impure. Although TMAH has virtually no odor when pure, samples often have a strongly fishy smell from the trimethylamine which is a common impurity. TMAH has numerous and diverse industrial and research applications.
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
الخصائص الكيميائية
البنية
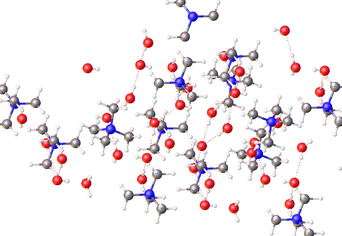
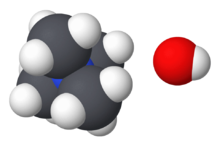
TMAH is most commonly encountered as an aqueous solution, in concentrations from ~2–25%, and less frequently as solutions in methanol. These solutions are identified by the CAS# 75-59-2. Several hydrates N(CH3)4OH·xH2O. have been crystallized.[3] These salts feature well separated Me4N+ cations and hydroxide anions. The hydroxide groups are linked by hydrogen bonds to the water of crystallization. Anhydrous TMAH has not been isolated.
التحضير
One of the earliest preparations is that of Walker and Johnston,[4] who made it by the salt metathesis reaction of tetramethylammonium chloride and potassium hydroxide in dry methanol, in which TMAH is soluble, but potassium chloride is not:
- NMe4+Cl− + KOH → NMe4+قالب:OH- + KCl
Where Me stands for the methyl group, CH3-.
This report also provides details for isolation of TMAH as its pentahydrate, noting the existence of a trihydrate, and emphasizes the avidity which even the former exhibits for atmospheric moisture and carbon dioxide. These authors reported a m.p. of 62–63 °C for the pentahydrate, and a solubility in water of 220 g/100 mL at 15 °C.
التفاعلات
- TMAH undergoes simple acid-base reactions with strong or weak acids to produce tetramethylammonium salts whose anion is derived from the acid. Illustrative is preparation of tetramethylammonium fluoride:[5]
- NMe4+قالب:OH- + HF → NMe4+F− + H2O
- Solutions of TMAH may be used to make other tetramethylammonium salts in a metathesis reaction with ammonium salts, whereby the anion is derived from the ammonium salt. The reaction is driven in the desired direction by evaporative removal of ammonia and water.[6] For example, tetramethylammonium thiocyanate may be made from ammonium thiocyanate, thus:
- NMe4+قالب:OH- + NH4+SCN− → NMe4+SCN− + NH3 + H2O
- TMAH, in common with many other TMA salts containing simple anions, decomposes on heating into trimethylamine.[7] Dimethyl ether is a major decomposition product rather than methanol.[8] The idealized equation is:
- 2 NMe4+قالب:OH- → 2 NMe3 + MeOMe + H2O
الخصائص
TMAH is a very strong base.[9]
الاستخدامات
One of the industrial uses of TMAH is for the anisotropic etching of silicon.[10] It is used as a basic solvent in the development of acidic photoresist in the photolithography process, and is highly effective in stripping photoresist. TMAH has some phase transfer catalyst properties, and is used as a surfactant in the synthesis of ferrofluid, to inhibit nanoparticle aggregation.
TMAH is the most common reagent currently used in thermochemolysis, an analytical technique involving both pyrolysis and chemical derivatization of the analyte.[11]
النقش المبتل متباين الخواص
TMAH belongs to the family of quaternary ammonium hydroxide (QAH) solutions and is commonly used to anisotropically etch silicon. Typical etching temperatures are between 70 and 90 °C and typical concentrations are 5–25 wt% TMAH in water. (100) silicon etch rates generally increase with temperature and increasing TMAH concentration. Etched silicon (100) surface roughness decreases with increasing TMAH concentration, and smooth surfaces can be obtained with 20% TMAH solutions. Etch rates are typically in the 0.1–1 micrometer per minute range.
Common masking materials for long etches in TMAH include silicon dioxide (LPCVD and thermal) and silicon nitride. Silicon nitride has a negligible etch rate in TMAH; the etch rate for silicon dioxide in TMAH varies with the quality of the film, but is generally on the order of 0.1 nm/minute.[10]
السمية
The tetramethylammonium ion [12] affects nerves and muscles, causing difficulties in breathing, muscular paralysis and possibly death.[13] It is structurally related to acetylcholine, an important neurotransmitter at both the neuromuscular junction and autonomic ganglia. This structural similarity is reflected in its mechanism of toxicity - it binds to and activates the nicotinic acetylcholine receptors, although they may become desensitized in the continued presence of the agonist. The action of tetramethylammonium is most pronounced in autonomic ganglia, and so tetramethylammonium is traditionally classed as a ganglion-stimulant drug.[14] The ganglionic effects may contribute to the deaths that have followed accidental industrial exposure, although the "chemical burns" induced by this strong base are also severe. There is evidence that poisoning can occur through skin-contact with concentrated solutions of TMAH.[15]
انظر أيضاً
- Quaternary ammonium cation
- كلوريد رباعي مثيل الأمونيوم Tetramethylammonium chloride
- رباعي مثيل الأمونيوم Tetramethylammonium
الهامش
- ^ أ ب ت ث Sigma-Aldrich Co., Tetramethylammonium hydroxide pentahydrate. Retrieved on 2015-04-06.
- ^ Mootz, Dietrich; Seidel, Reinhard (1990). "Polyhedral clathrate hydrates of a strong base: Phase relations and crystal structures in the system tetramethylammonium hydroxide-water". Journal of Inclusion Phenomena and Molecular Recognition in Chemistry. 8 (1–2): 139–157. doi:10.1007/BF01131293.
- ^ Hesse, W.; Jansen, M. (1991). "Polymorphie von Tetramethylammoniumhydroxid-Pentahydrat, NMe4OH.5H2O, und Kristallstrukturen der Raumtemperatur- und Tieftemperaturform". Zeitschrift für Anorganische und Allgemeine Chemie. 595: 115–130. doi:10.1002/zaac.19915950112.
- ^ Walker, J.; Johnston, J. (1905). "Tetramethylammonium hydroxide". J. Chem. Soc., Trans. 87: 955–961. doi:10.1039/ct9058700955.
- ^ "Syntheses, properties, and structures of anhydrous tetramethylammonium fluoride and its 1:1 adduct with trans-3-amino-2-butenenitrile". Journal of the American Chemical Society. 112 (21): 7619–7625. 1990. doi:10.1021/ja00177a025.
{{cite journal}}: Cite uses deprecated parameter|authors=(help) - ^ Markowitz, M. M. (1957). "A convenient method for preparation of quaternary ammonium salts". J. Org. Chem. 22: 983–984. doi:10.1021/jo01359a605.
- ^ Lawson, A. T.; Collie, N. (1888). "The action of heat on the salts of tetramethylammonium". J. Chem. Soc., Trans. 53: 1888. doi:10.1039/CT8885300624.
- ^ Musker, W. Kenneth. (1964). "A Reinvestigation of the Pyrolysis of Tetramethylammonium Hydroxide". Journal of the American Chemical Society. 86 (5): 960–961. doi:10.1021/ja01059a070.
- ^ Stewart, R.; O'Donnell, J. P. (1964). "Strongly basic systems: III.The H_ function for various solvent systems". Can. J. Chem. 42 (7): 1681–1693. doi:10.1139/v64-251.
- ^ أ ب Thong, J. T. L.; Choi, W. K.; Chong, C. W. (1997). "TMAH etching of silicon and the interaction of etching parameters". Sensors and Actuators A: Physical. 63 (3): 243–249. doi:10.1016/S0924-4247(97)80511-0.
- ^ Shadkami, F.; Helleur, R. (2010). "Recent applications in analytical thermochemolysis". J. Anal. Appl. Pyrol. 89: 2–16. doi:10.1016/j.jaap.2010.05.007.
- ^ Note that studies of the pharmacology and toxicology of TMA have typically been carried out using TMA halide salts - the hydroxide ion in TMAH is too destructive towards biological tissue.
- ^ Anthoni, U.; Bohlin, L.; Larsen, C.; Nielsen, P.; Nielsen, N. H.; Christophersen, C. (1989). "Tetramine: Occurrence in marine organisms and pharmacology". Toxicon. 27 (7): 707–716. doi:10.1016/0041-0101(89)90037-8. PMID 2675390.
- ^ Bowman, W.C. and Rand, M.J. (1980), "Peripheral Autonomic Cholinergic Mechanisms", in Textbook of Pharmacology 2nd Ed., Blackwell Scientific, Oxford 10.21
- ^ Lin, C.C.; et al. (2010). "Tetramethylammonium hydroxide poisoning". Clin. Toxicol. 48 (3): 213–217. doi:10.3109/15563651003627777. PMID 20230335.
