رباعي فلوروبورات النيتروسيل
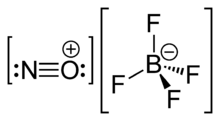
| |
| الأسماء | |
|---|---|
| اسم أيوپاك
nitrosonium tetrafluoroborate
| |
| أسماء أخرى
nitrosyl tetrafluoroborate
| |
| المُعرِّفات | |
| رقم CAS | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.035.148 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| الخصائص | |
| الصيغة الجزيئية | B4NO |
| كتلة مولية | 116.81 g mol-1 |
| المظهر | colourless crystalline solid |
| الكثافة | 2.185 g cm−3 |
| نقطة الانصهار | |
| قابلية الذوبان في الماء | decomposes |
ما لم يُذكر غير ذلك، البيانات المعطاة للمواد في حالاتهم العيارية (عند 25 °س [77 °ف]، 100 kPa). | |
| مراجع الجدول | |
Nitrosonium tetrafluoroborate, also called nitrosyl tetrafluoroborate, is a chemical compound with the chemical formula NOBF4. This colourless solid is used in organic synthesis as a nitrosating agent.[1]
NOBF4 is the nitrosonium salt of fluoroboric acid, and is composed of a nitrosonium cation, [NO]+, and a tetrafluoroborate anion, [BF4]−.
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Reactions
The dominant property of NOBF4 is the oxidizing power and electrophilic character of the nitrosonium cation. It forms colored charge transfer complexes with hexamethylbenzene and with 18-crown-6. The latter, a deep yellow species, provides a means to dissolve NOBF4 in dichloromethane.[2]
Nitrosonium tetrafluoroborate may be used to prepare metal salts of the type [MII(CH3CN)x][BF4]2 (M = Cr, Mn, Fe, Co, Ni, Cu). The nitrosonium cation acts as the oxidizer, itself being reduced to nitric oxide gas:[3]
- M + 2NOBF4 + xCH3CN → [M(CH3CN)x](BF4)2 + 2NO
With ferrocene the ferrocenium tetrafluoroborate is formed.[4]
In its infrared spectrum of this salt, νNO is a strong peak at 2387 cm−1.[5]
References
- ^ "A15806 Nitrosonium tetrafluoroborate, 98%". Alfa Aesar website. Retrieved 2010-09-04.
- ^ Lee, K. Y.; Kuchynka, D. J.; Kochi, Jay K. (1990). "Redox equilibria of the nitrosonium cation and of its nonbonded complexes". Inorganic Chemistry. 29 (21): 4196–4204. doi:10.1021/ic00346a008.
- ^ Heintz, Robert A.; Smith, Jennifer A.; Szalay, Paul S.; Weisgerber, Amy; Dunbar, Kim R. (August 2004). "11. Homoleptic Transition Metal Acetonitrile Cations with Tetrafluoroborate or Trifluoromethanesulfonate Anions". Inorg. Synth. 33: 75–83. doi:10.1002/0471224502.ch2. ISBN 978-0-471-46075-6.
- ^ Roger M. Nielson; George E. McManis; Lance K. Safford; Michael J. Weaver (1989). "Solvent and electrolyte effects on the kinetics of ferrocenium-ferrocene self-exchange. A reevaluation". J. Phys. Chem. 93 (5): 2152. doi:10.1021/j100342a086.
- ^ Sharp, D. W. A.; Thorley, J. (1963). "670. The Infrared Spectrum of the Nitrosonium Ion". Journal of the Chemical Society (Resumed): 3557. doi:10.1039/JR9630003557.