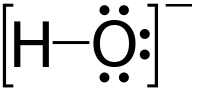زوج وحيد
الزوج الوحيد Lone pair هو زوج إلكترونات في ذرة بدون ترابط أو مشاركة مع ذرة أخرى. غالبا ما يكون له صفة قطبية سالبة لوجود كثافة الشحنة العالية. يستخدم هذا الزوج لعمل روابط تناسقية،[1] فمثلا عند تصنيع الهيدرونيوم، H3O+, تتواجد الأيونات عند إذابة الأحماض في الماء وتقوم عندها ذرة الأكسجين بإعطاء زوج وحيد لأيون الهيدروجين. Lone pairs are found in the outermost electron shell of atoms. They can be identified by using a Lewis structure. Electron pairs are therefore considered lone pairs if two electrons are paired but are not used in chemical bonding. Thus, the number of electrons in lone pairs plus the number of electrons in bonds equals the number of valence electrons around an atom.
Lone pair is a concept used in valence shell electron pair repulsion theory (VSEPR theory) which explains the shapes of molecules. They are also referred to in the chemistry of Lewis acids and bases. However, not all non-bonding pairs of electrons are considered by chemists to be lone pairs. Examples are the transition metals where the non-bonding pairs do not influence molecular geometry and are said to be stereochemically inactive. In molecular orbital theory (fully delocalized canonical orbitals or localized in some form), the concept of a lone pair is less distinct, as the correspondence between an orbital and components of a Lewis structure is often not straightforward. Nevertheless, occupied non-bonding orbitals (or orbitals of mostly nonbonding character) are frequently identified as lone pairs.
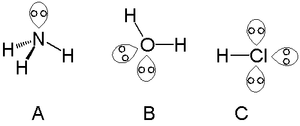
A single lone pair can be found with atoms in the nitrogen group, such as nitrogen in ammonia. Two lone pairs can be found with atoms in the chalcogen group, such as oxygen in water. The halogens can carry three lone pairs, such as in hydrogen chloride.
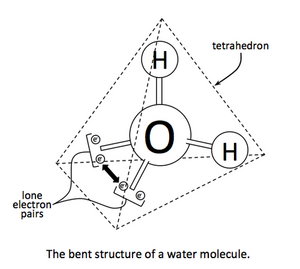
In VSEPR theory the electron pairs on the oxygen atom in water form the vertices of a tetrahedron with the lone pairs on two of the four vertices. The H–O–H bond angle is 104.5°, less than the 109° predicted for a tetrahedral angle, and this can be explained by a repulsive interaction between the lone pairs.[2][3][4]
Various computational criteria for the presence of lone pairs have been proposed. While electron density ρ(r) itself generally does not provide useful guidance in this regard, the Laplacian of the electron density is revealing, and one criterion for the location of the lone pair is where L(r) = –∇2ρ(r) is a local maximum. The minima of the electrostatic potential V(r) is another proposed criterion. Yet another considers the electron localization function (ELF).[5]
Angle changes
The pairs often exhibit a negative polar character with their high charge density and are located closer to the atomic nucleus on average compared to the bonding pair of electrons. The presence of a lone pair decreases the bond angle between the bonding pair of electrons, due to their high electric charge, which causes great repulsion between the electrons. They are also involved in the formation of a dative bond. For example, the creation of the hydronium (H3O+) ion occurs when acids are dissolved in water and is due to the oxygen atom donating a lone pair to the hydrogen ion.
This can be seen more clearly when looked at it in two more common molecules. For example, in carbon dioxide (CO2), the oxygen atoms are on opposite sides of the carbon (linear molecular geometry), whereas in water (H2O) the angle between the hydrogen atoms is 104.5° (bent molecular geometry). The repulsive force of the oxygen atom's lone pairs pushes the hydrogens further apart, until the forces of all electrons on the hydrogen atom are in equilibrium. This is an illustration of the VSEPR theory.
Dipole moments
Lone pairs can contribute to a molecule's dipole moment. NH3 has a dipole moment of 1.42 D. As the electronegativity of nitrogen (3.04) is greater than that of hydrogen (2.2) the result is that the N-H bonds are polar with a net negative charge on the nitrogen atom and a smaller net positive charge on the hydrogen atoms. There is also a dipole associated with the lone pair and this reinforces the contribution made by the polar covalent N-H bonds to ammonia's dipole moment. In contrast to NH3, NF3 has a much lower dipole moment of 0.234 D. Fluorine is more electronegative than nitrogen and the polarity of the N-F bonds is opposite to that of the N-H bonds in ammonia, so that the dipole due to the lone pair opposes the N-F bond dipoles, resulting in a low molecular dipole moment.[6]
Stereogenic lone pairs

|
⇌ | 
|
| Inversion of a generic organic amine molecule at nitrogen | ||
A lone pair can contribute to the existence of chirality in a molecule, when three other groups attached to an atom all differ. The effect is seen in certain amines, phosphines,[7] sulfonium and oxonium ions, sulfoxides, and even carbanions.
The resolution of enantiomers where the stereogenic center is an amine is usually precluded because the energy barrier for nitrogen inversion at the stereo center is low, which allow the two stereoisomers to rapidly interconvert at room temperature. As a result, such chiral amines cannot be resolved, unless the amine's groups are constrained in a cyclic structure (such as in Tröger's base).
Unusual lone pairs
أوصاف مختلفة للعديد من الأزواج الوحيدة
In elementary chemistry courses, the lone pairs of water are described as "rabbit ears": two equivalent electron pairs of approximately sp3 hybridization, while the HOH bond angle is 104.5°, slightly smaller than the ideal tetrahedral angle of arccos(–1/3) ≈ 109.47°. The smaller bond angle is rationalized by VSEPR theory by ascribing a larger space requirement for the two identical lone pairs compared to the two bonding pairs. In more advanced courses, an alternative explanation for this phenomenon considers the greater stability of orbitals with excess s character using the theory of isovalent hybridization, in which bonds and lone pairs can be constructed with spx hybrids wherein nonintegral values of x are allowed, so long as the total amount of s and p character is conserved (one s and three p orbitals in the case of second-row p-block elements).
انظر أيضاً
- Coordination complex
- HOMO and LUMO (highest occupied molecular orbital and lowest unoccupied molecular orbital)
- Inert-pair effect
- Ligand
- Shared pair
المصادر
- ^ IUPAC Gold Book definition: lone (electron) pair
- ^ Fox, M.A.; Whitesell, J.K. (2004). Organic Chemistry. Jones and Bartlett Publishers. ISBN 978-0-7637-2197-8. Retrieved 5 May 2021.
- ^ McMurry, J. (2000). Organic Chemistry 5th Ed. Ceneage Learning India Pvt Limited. ISBN 978-81-315-0039-2. Retrieved 5 May 2021.
- ^ Lee, J.D. (1968). Concise Inorganic Chemistry. Student's paperback edition. Van Nostrand. Retrieved 5 May 2021.
- ^ Kumar, Anmol; Gadre, Shridhar R.; Mohan, Neetha; Suresh, Cherumuttathu H. (2014-01-06). "Lone Pairs: An Electrostatic Viewpoint". The Journal of Physical Chemistry A (in الإنجليزية). 118 (2): 526–532. Bibcode:2014JPCA..118..526K. doi:10.1021/jp4117003. ISSN 1089-5639. PMID 24372481.
- ^ قالب:Housecroft2nd
- ^ Quin, L. D. (2000). A Guide to Organophosphorus Chemistry, LOCATION: John Wiley & Sons. ISBN 0471318248.