أوپيورفين
| |
| الأسماء | |
|---|---|
| اسم أيوپاك
(2S,5S,8S,11S,14S)-14,17-diamino-8-benzyl-2,11-bis(3-guanidinopropyl)-5-(hydroxymethyl)-4,7,10,13,17-pentaoxo-3,6,9,12-tetraazaheptadecan-1-oic acid
| |
| أسماء أخرى
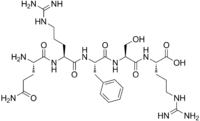
Gln-Arg-Phe-Ser-Arg; L-Glutaminyl-L-arginyl-L-phenylalanyl-L-seryl-L-arginine
| |
| المُعرِّفات | |
| رقم CAS | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
CompTox Dashboard (EPA)
|
|
| |
| |
| الخصائص | |
| الصيغة الجزيئية | C29H48N12O8 |
| كتلة مولية | 692.72 g mol-1 |
ما لم يُذكر غير ذلك، البيانات المعطاة للمواد في حالاتهم العيارية (عند 25 °س [77 °ف]، 100 kPa). | |
| مراجع الجدول | |
اوپيورفين Opiorphin هو مركب كيميائي endogenous عـُزِل لأول مرة من اللعاب البشري. الأبحاث الأولية مع الفئران تبين أن المركب له تأثير مسكن للألم أكبر من تأثير المورفين.[2] ويعمل بإيقاف التكسر العادي للإنكفالينات، وهي opioids المسكنة للألم الطبيعية في الحبل العصبي. وهو جزء بسيط نسبياً مكوّن من خمس پپتيدات متعددة لأحماض أمينية، Gln-Arg-Phe-Ser-Arg.[3][4][5][6][7][8][9][10]
Opiorphin pentapeptide originates from the N-terminal region of the protein PROL1 (proline-rich, lacrimal 1).[3] Opiorphin inhibits three proteases: neutral ecto-endopeptidase (MME), ecto-aminopeptidase N (ANPEP)[3] and perhaps also a dipeptidyl peptidase DPP3.[8] Such action extends the duration of enkephalin effect where the natural pain killers are released physiologically in response to specific potentially painful stimuli, in contrast with administration of narcotics, which floods the entire body and causes many undesirable adverse reactions, including addiction liability and constipation.[11][12] In addition, opiorphin may exert anti-depressive action.[13][14]
الاستخدام العلاجي للأوپيورفين في البشر سيتطلب تعديل الجزيء لتجنب تحلله السريع في الأمعاء واختراقه الضئيل لـ حاجز الدم-المخ.[11][12]
انظر أيضاً
الهامش
- ^ Opiorphin at Sigma-Aldrich
- ^ Rougeot C, Robert F, Menz L, Bisson JF, Messaoudi M (August 2010). "Systemically active human opiorphin is a potent yet non-addictive analgesic without drug tolerance effects". J. Physiol. Pharmacol. 61 (4): 483–90. PMID 20814077.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ أ ب ت Dickinson DP, Thiesse M (April 1996). "cDNA cloning of an abundant human lacrimal gland mRNA encoding a novel tear protein". Curr. Eye Res. 15 (4): 377–86. doi:10.3109/02713689608995828. PMID 8670737.
- ^ Andy Coghlan (November 13, 2006). "Natural-born painkiller found in human saliva". New Scientist.
- ^ "Natural chemical 'beats morphine'". BBC News. November 14, 2006.
- ^ Mary Beckman (November 13, 2006). "Prolonging Painkillers". ScienceNOW.
- ^ Stanović S, Boranić M, Petrovecki M; et al. (2000). "Thiorphan, an inhibitor of neutral endopeptidase/enkephalinase (CD10/CALLA) enhances cell proliferation in bone marrow cultures of patients with acute leukemia in remission". Haematologia (Budap). 30 (1): 1–10. doi:10.1163/15685590051129814. PMID 10841318.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ أ ب Thanawala V, Kadam VJ, Ghosh R (October 2008). "Enkephalinase inhibitors: potential agents for the management of pain". Curr Drug Targets. 9 (10): 887–94. doi:10.2174/138945008785909356. PMID 18855623.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Davies KP (March 2009). "The role of opiorphins (endogenous neutral endopeptidase inhibitors) in urogenital smooth muscle biology". J Sex Med. 6 Suppl 3: 286–91. doi:10.1111/j.1743-6109.2008.01186.x. PMC 2864530. PMID 19267851.
- ^ Tian XZ, Chen J, Xiong W, He T, Chen Q (July 2009). "Effects and underlying mechanisms of human opiorphin on colonic motility and nociception in mice". Peptides. 30 (7): 1348–54. doi:10.1016/j.peptides.2009.04.002. PMID 19442408.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ أ ب Rougeot C, Robert F, Menz L, Bisson JF, Messaoudi M (August 2010). "Systemically active human opiorphin is a potent yet non-addictive analgesic without drug tolerance effects" (PDF). J. Physiol. Pharmacol. 61 (4): 483–90. PMID 20814077.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ أ ب Popik P, Kamysz E, Kreczko J, Wróbel M (November 2010). "Human opiorphin: the lack of physiological dependence, tolerance to antinociceptive effects and abuse liability in laboratory mice". Behav. Brain Res. 213 (1): 88–93. doi:10.1016/j.bbr.2010.04.045. PMID 20438769.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Javelot H, Messaoudi M, Garnier S, Rougeot C (June 2010). "Human opiorphin is a naturally occurring antidepressant acting selectively on enkephalin-dependent delta-opioid pathways" (PDF). J. Physiol. Pharmacol. 61 (3): 355–62. PMID 20610867.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Yang QZ, Lu SS, Tian XZ, Yang AM, Ge WW, Chen Q (February 2011). "The antidepressant-like effect of human opiorphin via opioid-dependent pathways in mice". Neurosci. Lett. 489 (2): 131–5. doi:10.1016/j.neulet.2010.12.002. PMID 21145938.
{{cite journal}}: CS1 maint: multiple names: authors list (link)
- Articles with changed EBI identifier
- Articles with changed ChemSpider identifier
- Articles with changed InChI identifier
- Articles containing unverified chemical infoboxes
- Chembox image size set
- Chemical articles with unknown parameter in Chembox
- Short description is different from Wikidata
- مسكنات ألم
- پپتيدات عصبية
- مثبطات الإنكفالينيز
- پپتيدات شبه أفيونية